MedtecLIVE 2023
Thank you for your time and the pleasant conversation at MedTecLIVE 2023.
Down below you will find all relevant information and downloads about our software solutions for medical devices.
infoteam Software Group has been implementing specific software solutions for 40 years, including for its customers in the life science sector. infoteam is certified according to ISO 9001, ISO 13485 as well as ISO 27001 and has its own development process that supports IVDR-, MDR- and FDA-compliant software development for medical devices of classes I to III as well as safety classes A to C and meets the relevant requirements of IEC 62304.
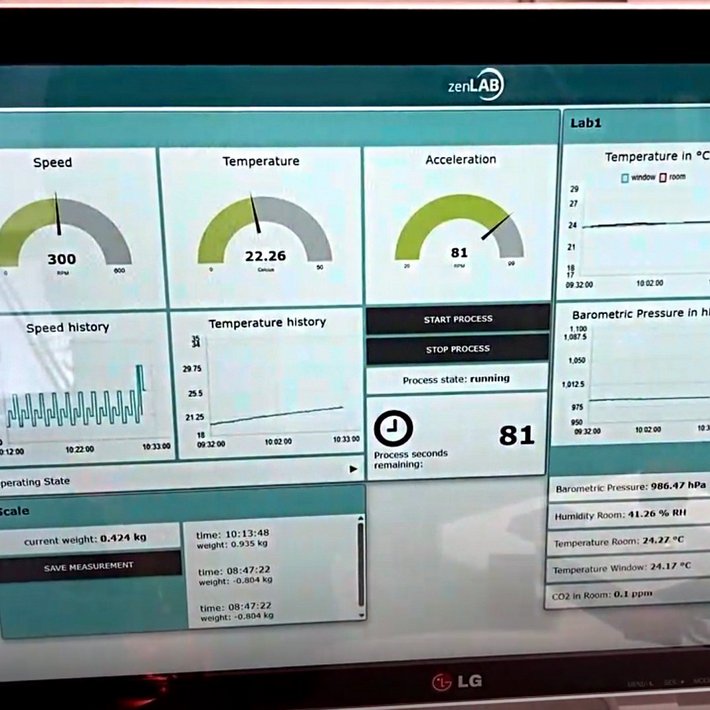
The life science portfolio includes the development of embedded software for medical devices, application software, medical apps (incl. DiGA) as well as data analysis and AI solutions - always over the entire life cycle, in accordance with the state of the art and taking security requirements into account.
The infoteam Software Group employs more than 300 people in Europe, its headquarters are in the metropolitan region of Nuremberg. infoteam has received the New Work Star Award and is one of the TOP 100 most innovative medium-sized companies in Germany.
If you have any questions, our experts will be happy to help and advise you. We look forward to hearing from you.

E-Mail:
s.buettner@infoteam.de
Phone:
+49 9131 78 00-280
I am looking for the right contact in our company for your topic and look forward to hearing from you.
Make an appointment online